COVID-19 vaccine: UK regulators warn people with history of ‘significant’ allergic reactions not to have Pfizer/BioNTech jab.
By Chris Robertson, news reporter
UK regulators have issued a warning that people who have a history of “significant” allergic reactions should not currently receive the Pfizer/BioNTech vaccine.
It comes after two NHS staff members who had the jab yesterday experienced allergic reactions.
They developed “anaphylactoid reaction” symptoms shortly after being injected, and are now understood to be recovering.
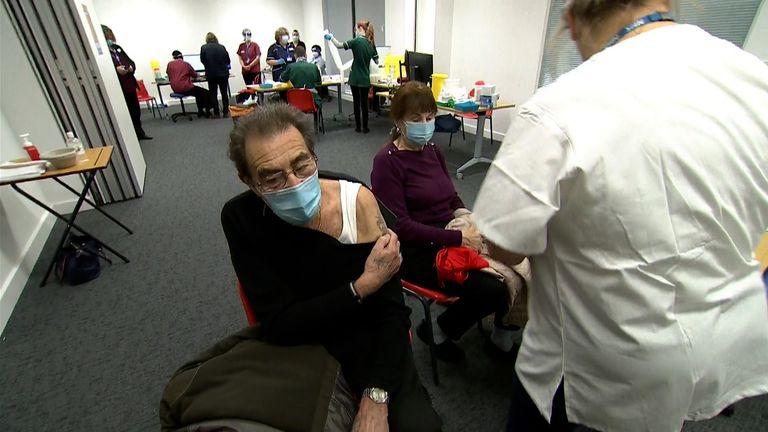
Both have a significant history of allergies and have to carry adrenaline.
The pair are among thousands of people who received the first dose of the jab yesterday, as the biggest vaccination programme in the UK’s history got under way.
The UK became the first country in the world to approve the Pfizer vaccine last week, with care home workers, hospital patients and NHS staff, among others, getting the first jabs.
The Medicines and Healthcare products Regulatory Agency (MHRA) has given precautionary advice to NHS trusts that anyone who has a history of “significant” allergic reactions to medicines, food or vaccines should not receive the vaccine.
It added that anyone who has a significant reaction to the first jab should not have the second dose.
NHS England said all trusts involved with the vaccination programme have been informed and from Wednesday, all patients scheduled to receive the Pfizer vaccine will be asked if they have a history of allergic reactions.
A spokeswoman for Pfizer said its vaccine was “well tolerated” during the trials, with “no serious safety concerns”.
In a statement, Pfizer UK said: “We have been advised by MHRA of two yellow card reports that may be associated with allergic reaction due to administration of the COVID-19 BNT162b2 vaccine.
“As a precautionary measure, the MHRA has issued temporary guidance to the NHS while it conducts an investigation in order to fully understand each case and its causes. Pfizer and BioNTech are supporting the MHRA in the investigation.
“In the pivotal phase 3 clinical trial, this vaccine was generally well tolerated with no serious safety concerns reported by the independent Data Monitoring Committee. The trial has enrolled over 44,000 participants to date, over 42,000 of whom have received a second vaccination.”
Sky’s Thomas Moore says: “The regulator is likely to investigate what may have caused the reactions. The vaccine contains RNA, the genetic material of the coronavirus, as well as a liquid to stabilise and dilute it during the injection.
“Some vaccines already carry health warnings for people prone to allergic reactions – people who have an egg allergy are told not to have certain brands of flu vaccine because they are grown in eggs.
“Many scientists believe RNA vaccines are ‘purer’ and less likely to cause side effects.”
Dr June Raine, the head of the MHRA, told a joint select committee hearing that “real-time vigilance” would continue even now the vaccine had been deployed.
“Even last evening we were looking at two case reports of allergic reactions.
“We know from the extensive clinical trials that this was not a feature, but if we need to strengthen our advice now that we have had this experience in vulnerable populations, the groups selected as a priority, we get that advice out immediately.”














